|
The first and second laws of thermodynamics Thus, modeling and predicting the folding process, and its final structure, is an important problem for thermodynamics to help solve. The enzyme may require active sites, or pockets, to physically hold the reactants of a biochemical reaction, so the correct folding into this final structure is essential for the function of the enzyme molecule. But if, for example, the protein is going to act as an enzyme, then the final structure is critical for the molecule to perform its catalytic function. After protein synthesis has occurred in the cell, the protein macromolecule has completed its chemical synthesis. Interactions with the environment are important considerations when describing the self-assembly of a molecule, since satisfying the increase in entropy of the second law depends on the environment “absorbing” the entropy increase, while the self-assembly process itself can lead to a more ordered molecule, or a local decrease in entropy.Īn important example of molecular self-assembly in biology is protein folding.  
The thermal states of the system depend on the interactions with the immediate surroundings, as do any entropy changes. Sprackling calls the changing state variables, such as temperature changes, the paths of the process 2. This is an ideal condition, and in real microscopic processes equilibrium states blend together, and the state variables change in a continuous way. In thermodynamic terms, the process of self-assembly passes from one equilibrium state to another equilibrium state through infinitesimally small steps. The microscopic process that is being described is molecular self-assembly. In this study of molecular self-assembly, systems are biological systems, such as the interior of a cell of an organism, and the macromolecules constitute a subsystem. In thermal physics, the ideal system is an isolated whole which contains the matter being studied, and is assumed to be at equilibrium, with state variables, such as temperature, constant. Michael Sprackling in the book “Thermal Physics” stresses the importance of defining the system and processes that science is describing 1. An important point to note is that when a macromolecule self-assembles, such as the folding of a large protein molecule, the molecule becomes more ordered and the local entropy decreases, however, the entropy of the entire system (molecule and environment) must increase by the second law. To better understand the molecular self-assembly process, and statistical models of this process, I will be focusing on entropy and the second law of thermodynamics. 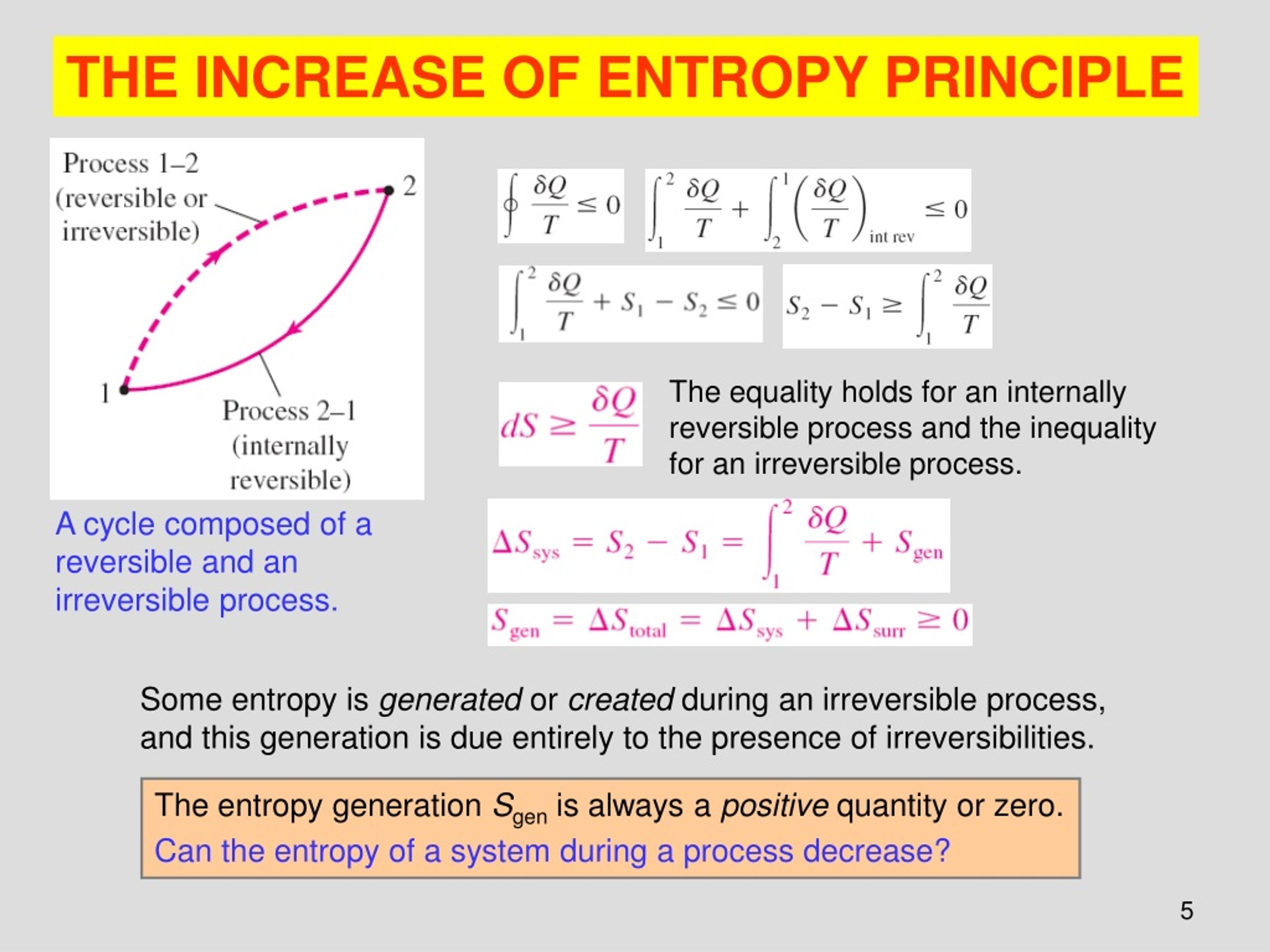
This system must obey the statistical laws of thermodynamics: the first law of thermodynamics has to do with the conservation of energy and the second law has to do with increasing entropy with time. The macromolecule represents a thermodynamic subsystem, and together with its immediate environment, constitutes an idealized thermodynamic system. 
The microscopic activity for this study is the self-assembly of large macromolecules. The macroscopic properties might be the temperature, representing the system's internal energy, the volume, which is inversely related to the pressure, or the entropy, a measure of the system's disorder. Statistical Physics is interested in understanding the macroscopic properties of microscopic activities.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
